 Dr. Jewell had the honor of presenting the 24 month outcome data for the use of a new biologic textile that was studied for soft tissue support during breast reconstruction after mastectomy. He attended the ORBS Symposia, an international oncoplastic reconstructive breast surgery meeting that was held in Nottingham, England. There was a podium presentation of the 24 month outcome data where Seri Surgical Scaffold was used to provide soft tissue support during breast reconstruction. The outcome data came from multi-center grouping of 9 researchers and approximately 139 women who underwent breast reconstruction following mastectomy. Seri Surgical Scaffold (Allergan), a new biologic textile derived from silk was utilized for soft tissue support during the reconstruction. This study was designed to evaluate the ability of Seri Surgical Scaffold to provide durable soft tissue support during breast reconstruction.
Dr. Jewell had the honor of presenting the 24 month outcome data for the use of a new biologic textile that was studied for soft tissue support during breast reconstruction after mastectomy. He attended the ORBS Symposia, an international oncoplastic reconstructive breast surgery meeting that was held in Nottingham, England. There was a podium presentation of the 24 month outcome data where Seri Surgical Scaffold was used to provide soft tissue support during breast reconstruction. The outcome data came from multi-center grouping of 9 researchers and approximately 139 women who underwent breast reconstruction following mastectomy. Seri Surgical Scaffold (Allergan), a new biologic textile derived from silk was utilized for soft tissue support during the reconstruction. This study was designed to evaluate the ability of Seri Surgical Scaffold to provide durable soft tissue support during breast reconstruction.
 Seri Surgical Scaffold was developed at Tufts University and is FDA-approved. It is made from ultra-pure silk and is placed during reconstructive breast surgery to provide soft tissue support in the lower part of the breast where tissue has been removed. It can be used with a tissue expander of in direct-to-implant reconstructions. Seri has a unique interaction with the body’s tissues, where new collagen is produced and the textile is absorbed over about 12 months’ time. According to Dr. Jewell, this is the optimal approach for generating new tissue that provides support versus relying on plastic mesh or tissues from cadavers or pigs.
Seri Surgical Scaffold was developed at Tufts University and is FDA-approved. It is made from ultra-pure silk and is placed during reconstructive breast surgery to provide soft tissue support in the lower part of the breast where tissue has been removed. It can be used with a tissue expander of in direct-to-implant reconstructions. Seri has a unique interaction with the body’s tissues, where new collagen is produced and the textile is absorbed over about 12 months’ time. According to Dr. Jewell, this is the optimal approach for generating new tissue that provides support versus relying on plastic mesh or tissues from cadavers or pigs.
 >The outcome data that was presented at the ORBS meeting by Dr. Jewell demonstrated that women had a high degree of satisfaction with the quality of their reconstruction when Seri Surgical Scaffold was used. The safety profile of Seri was also better than reported outcome studies that use cadaver or porcine-sourced tissue in a similar fashion. According to Dr.
>The outcome data that was presented at the ORBS meeting by Dr. Jewell demonstrated that women had a high degree of satisfaction with the quality of their reconstruction when Seri Surgical Scaffold was used. The safety profile of Seri was also better than reported outcome studies that use cadaver or porcine-sourced tissue in a similar fashion. According to Dr.
Jewell’s presentation, the incidence of serious adverse events such as infection with Seri was 2.3% versus 15% that was reported in a similar study that used cadaver-sourced tissue.
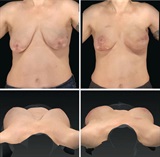 Dr. Jewell and the other researchers in the study noted excellent stability of the breast mound during the 24 month period of the study, according to 3-D imaging and analysis. There was no need for re-operation to correct soft tissue stretch that frequently happens when cadaver-sourced tissue is used in this capacity. Dr. Jewell believes that the Seri Surgical Scaffold provides a durable, yet supple tissue that resists stretching over time, useful in the breast and abdominal wall areas. This represents better options for women needing breast reconstruction after mastectomy.
Dr. Jewell and the other researchers in the study noted excellent stability of the breast mound during the 24 month period of the study, according to 3-D imaging and analysis. There was no need for re-operation to correct soft tissue stretch that frequently happens when cadaver-sourced tissue is used in this capacity. Dr. Jewell believes that the Seri Surgical Scaffold provides a durable, yet supple tissue that resists stretching over time, useful in the breast and abdominal wall areas. This represents better options for women needing breast reconstruction after mastectomy.


Leave a Reply