Mark L. Jewell, MD
Alireza “Reza” Najafian, MD
1200 Executive Parkway Suite 360
Eugene, OR 97401
Phone: (541) 683-3234
Fax: (541) 683-8610
Monday – Thursday: 9 a.m.–5 p.m.
Friday: 9 a.m.–2 p.m.
Breast Implant Options
Dr. Mark Jewell and Dr. Alireza “Reza” Najafian create customized breast augmentation results by offering a variety of breast implants to patients from Eugene, Salem, Bend, and throughout Oregon. The breast implant options available today include the newest generation of highly filled cohesive silicone gel implants from all 3 companies that make implants.
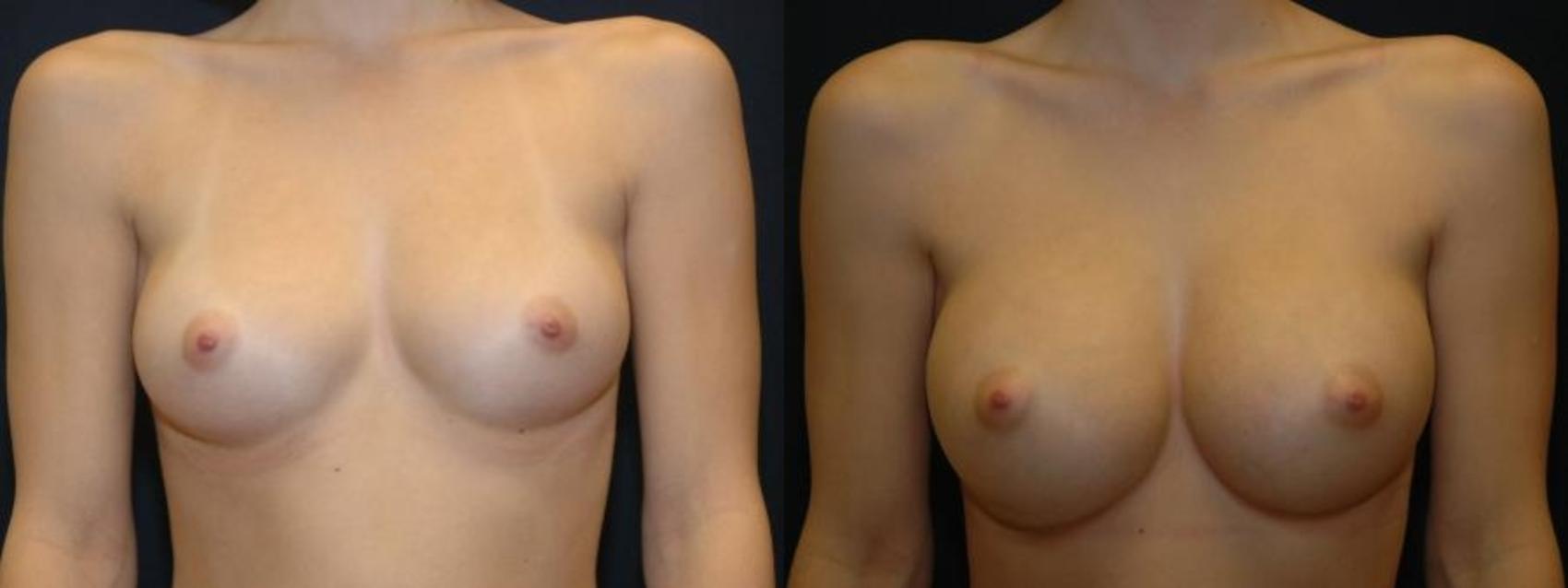
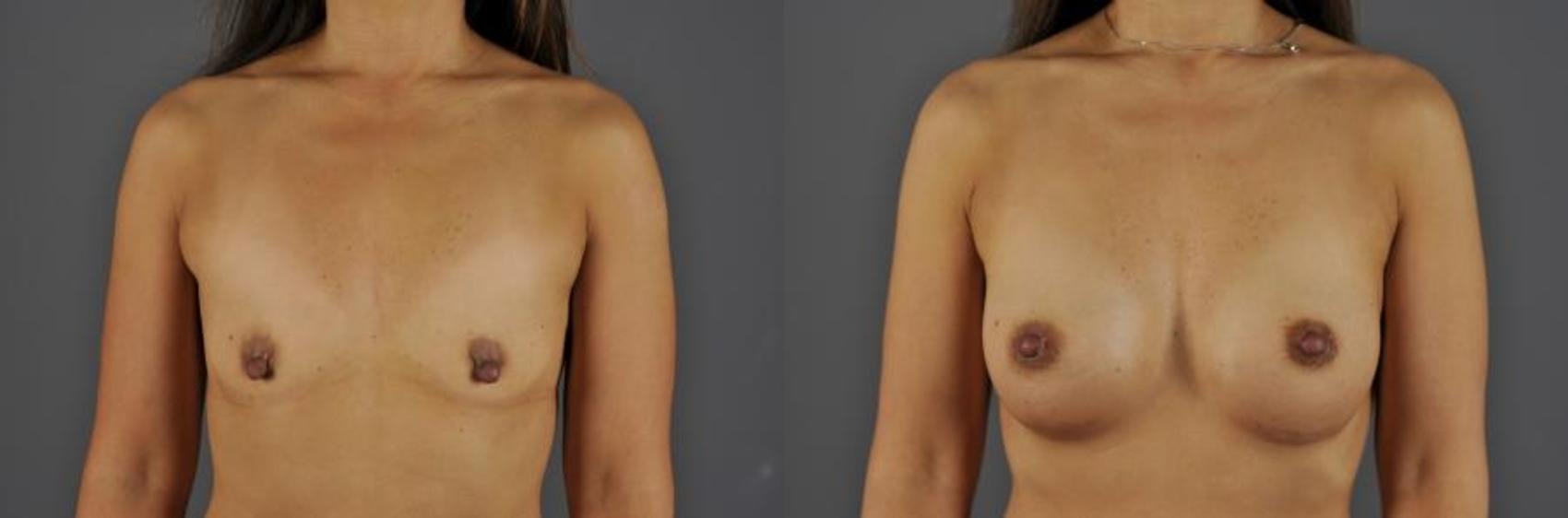
Breast Augmentation
Before & After Photos
WARNING:
This feature contains nudity. Please click OK to confirm you are at least 18 years of age and are not offended by such material.
Choosing Breast Implants
Modern breast augmentation is an individually customized operation designed to produce beautiful breasts, with approximately 45% of the breast volume in the upper breast and 55% in the lower. For some patients whose breasts lack shape, our surgeons may recommend an anatomically shaped implant. Most other patients benefit from a round implant that fills the breast envelope.
Several factors influence the choice of breast implants, including physical measurements of the patient’s breasts, the size of breasts she wants, and the location of the implants within the breasts. Dr. Jewell and Dr. Najafian use both the highly filled, round implants that are part of the Allergan Natrelle INSPIRA® product line and the form-stable, anatomically shaped Allergan Natrelle® 410. Dr. Jewell was among the original group of investigators conducting the FDA study that brought the Allergan Natrelle 410 implants to market. Additionally, Dr. Jewell’s experience with the Allergan Natrelle INSPIRA implants began years ago when he was an international professor of plastic surgery.
The Allergan Natrelle INSPIRA is available in 3 different silicone gel formulations and various sizes. This means Dr. Jewell and Dr. Najafian can match a specific implant to the patient’s breast measurements and her size expectations. The different consistencies of silicone gel that fill the implants enable him to adjust the fullness in the upper breast.
Saline-filled implants are still available, but few patients prefer them. The most recent national statistics show only about 15% of breast augmentation procedures use saline implants. Saline implants often develop visible ripples or wrinkles and feel like the breast is filled with a water balloon. They also tend to sag more often than silicone gel implants because gravity pulls down on the liquid solution.
Our Exceptional Providers
Dr. Jewell and Dr. Najafian combine artistry and surgical expertise to create customized results tailored to each patient's unique needs.
Implant Placement
Part of the decision-making process for breast augmentation involves implant placement. In general, implants can be placed either behind the breast tissue and either in front of the chest muscle (pectoralis major), or behind the muscle.
Our surgeons consider several factors before recommending a placement, including the amount of existing breast tissue, how much the breasts sag, the patient’s activity level, and how much she’s concerned about distorting the shape of the breasts when the chest muscles are tightened. According to Dr. Jewell’s published outcome data, patients can achieve good outcomes from breast implants placed in front of the chest muscle (sub-fascial) if they have ample breast tissue to cover the implant.
This appeals to athletic women, patients with some breast sagginess but who do not need a breast lift, and those who do not want to have their breasts move when they tighten their chest muscles. Sub-fascial implant placement keeps the implant and the breast tissue together and prevents the late-term “waterfall” deformity, which involves breast tissue falling from an implant placed behind the muscle (submuscular).
Some patients with very limited amounts of existing breast tissue may require using the chest muscle to cover the top of the implant for the best aesthetic outcome. It also minimizes the risk of implant malposition in athletic women, whose vigorous movements involving chest muscles can cause this problem
Which Implants Are Best for You?
Dr. Jewell and Dr. Najafian believe that the Allergan Natrelle Inspira line of implants can be placed either in front of the chest muscle (sub-fascial), provided there is ample breast tissue to cover the implant, or partially behind the muscle (bi-planar location) if there is thin tissue. They prefer the Allergan Natrelle Inspira with a smooth surface.
The Allergan 410 is a shaped implant with a tapered upper edge to create the appearance of a natural breast. It can be placed either in front of the chest muscle (sub-fascial) or behind (bi-planar). One difference is that the Allergan Natrelle 410’s textured surface helps hold it in place and minimizes the risk that the implant will rotate. According to Dr. Jewell, textured implant surfaces are useful yet must be used with caution to prevent bacterial surface contamination at the time of insertion.
Minimizing Risks of BIA-ALCL
All types of implanted medical devices are at risk for bacterial surface contamination during surgery (biofilm). This can contribute to capsular contracture. For patients with textured implants, it is also associated with the rare occurrence of breast implant-associated anaplastic large cell lymphoma (BIA-ALCL). This condition occurs around the implant, years later. BIA-ALCL has not been seen in patients who have smooth surface implants.
Our surgeons minimize the risk of these complications by using specific surgical techniques and processes that help prevent implant surface contamination with biofilm. They combine precise surgical techniques with antibiotics and irrigate the implant pocket with Betadine (iodine-based irrigation). They also use the Keller Funnel® to insert implants, a device that limits the handling of the implants. Dr. Jewell believes that the use of an iodine-containing irrigation solution is important because it kills the type of bacteria that is thought to cause BIA-ALCL.
Dr. Jewell’s clinical outcome data demonstrates that these measures are effective. To date, after 41 years of using breast implants, he has never had an infection in primary breast augmentation or revision surgery. He, along with 7 other plastic surgeons, published their outcome data involving approximately 42,000 implants in 21,650 patients that had a mean follow-up of 11.7 years where techniques were used to minimize biofilm contamination at the time of surgery. To date, there have not been any reported cases of BIA-ALCL. While the incidence of BIA-ALCL is remote, if patients are concerned about this, they should consider smooth surface implants.
If you’re looking in Eugene, Oregon, for a breast augmentation surgeon, request a consultation with Dr. Jewell or Dr. Najafian using the online form or call us at (541) 683-3234 to schedule an appointment.
Jewell Med Spa & Specials
It’s never too early to begin the journey toward healthier, youthful looking skin.
Learn More